RECENT SCIENTIFIC RESEARCH IN PAPER CONSERVATIONDIANNE VAN DER REYDEN
ABSTRACT—Scientific research in paper conservation since 1988 is discussed and summarized. Research on chemical, physical, and optical properties of paper and on the aging of paper is outlined. A synopsis is given of recent findings in paper conservation concerned with washing, bleaching, solvents, enzymes, and sizing. 1 INTRODUCTIONIn 1988, the first American Institute for Conservation Book and Paper Specialty Group Update Session reviewed scientific research in five important areas of paper conservation: encapsulation, fumigation, washing, deacidification, and bleaching (Kruth 1988). Research published since then continues to add to the body of knowledge about these and other treatments, and to shed light on the relationship of treatments to paper properties and the aging of paper (Robb 1991). Paper conservators are increasingly concerned about the long-term effects of treatments not only on media, but on the chemical and physical properties of all the components of the paper composite, including the fibers, fibrils, and cellulose polymer chain (fig. 1). This review summarizes scientific research pertinent to these concerns. To provide the context for evaluation of paper conservation research, information regarding paper properties and the aging of paper is outlined first, followed by a synopsis of recent findings about paper conservation treatments involving washing, bleaching, solvents, enzymes, and sizing. The information in this article is highly selective and condensed, representing only a small portion of the research data. The reader is urged to consult the cited references for more details and additional findings.
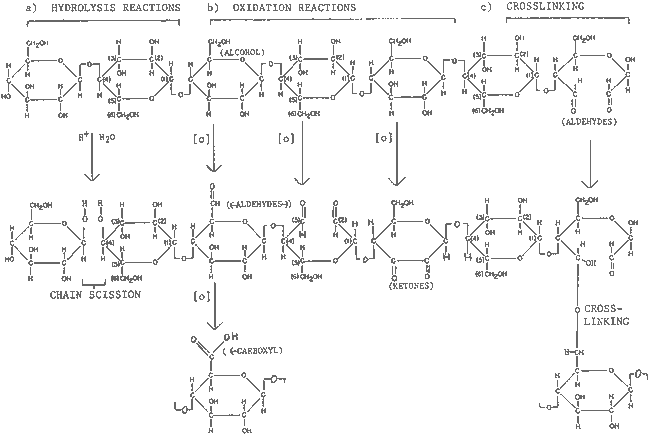
2 PAPER PROPERTIESBurgess and Binnie (1990) advise that “The evaluation of scientific results and recommendations in conservation literature is greatly aided by a good understanding of the rationale behind the selection of test materials and methods.” An understanding of the rationale behind research design is enhanced by a knowledge of paper properties and their reaction to relative humidity, temperature, radiation, and pollution. These environmental factors initiate degradation mechanisms of hydrolysis, oxidation, and crosslinking (Table 1). These degradation mechanisms can be detected by various tests (Table 2), which measure the chemical, physical, and optical properties of paper. The following summary presents a few basic terms and concepts relevant to these properties. (Optical properties are a type of physical property and can be used to monitor chemical change. They are covered separately here for purposes of organization.)
2.1 CHEMICAL PROPERTIESChemical properties of a paper sheet are derived primarily from variables of furnish or pulp components and from formation or manufacturing processes (Dwan 1987). Furnish variables include the fiber type; pulp stock; refining techniques such as beating or bleaching; and composition including fillers, sizes, coatings, and colorants (Priest 1989a). Formation variables include basis weight, calendaring, and manufacture variables such as speed, drainage, drying, and fiber bonding. Each variable in turn has distinct properties. For example, the pulp stock from which a paper is made has specific properties, including crystallinity, fiber bonding, viscosity, and drying, which affect the nature of the final sheet. Refining techniques such as beating affect fibrillation, swelling, hydration, and water retention. On the molecular level, cellulose, the primary and most stable component of paper fibers (fig. 1), has properties imposed by its structure (fig. 2), which creates amorphous and crystalline regions in fibrils (Smith 1991). The amorphous regions are random, flexible, and water accessible (fig. 3), while the crystalline regions are ordered, rigid, inert, and relatively impermeable to water (Kremer and Tabb 1990). Examples of chemical properties of paper (table 2) and their respective test methods (Waterhouse 1988; Wilson and Parks 1979) are listed below.
It is important to note that it is the noncellulosic components in some papers that are the most reactive, as in the case of lignin-containing papers. Ironically, this very instability makes such papers the most difficult to monitor and measure chemically. 2.2 PHYSICAL PROPERTIESPhysical properties of paper sheets are determined by basis weight, thickness, density, two-sidedness, smoothness, permeability, handle, rigidity, roughness, and porosity, which in turn affect sheet properties of softness, hardness, compressibility, dimensional stability, curl, and strength (Casey 1980; Bolam 1962). Attention tends to focus on strength measurements since “strength properties can … serve as indications of the permanence of paper, even when the nature of the chemical changes responsible for the deterioration remains unknown … [and] can also be used for evaluating the effectiveness of treatments for aging” (Caulfield and Gunderson 1988). The following are some strength, or mechanical, properties and examples of respective test methods.
2.3 OPTICAL PROPERTIESAs mentioned, optical properties are physical properties which conservators often monitor as an indication of chemical changes. Optical properties of paper include color, brightness, whiteness, reflectance, gloss, opacity, transparency or transmittance of light, and fluorescence (Mark 1983). Measurements are made with reflectometers, spectrophotometers, colorimeters, and goniometers. Findings may be reported in various modes including percent reflectance (figs. 6a, 8a), CieL∗a∗b∗ (fig. 9e) and Kubelka-Munk scattering and absorption coefficients (Berndt 1989; Burgess and Binnie 1991).
Paper properties are interdependent. Interpretation of test data is complicated by the fact that measurements represent “a combination of such factors as flexibility, bonding strength, and fiber strength … which are dependent on type of fibers, length and thickness of fibers, imperfections in fibers, flexibility, pattern of fiber network, number of bonds, strength of the individual bonds, the weight of the paper, the apparent density, the moisture content, and many other factors” (Casey 1980). Properties are in turn affected individually and collectively by environmental parameters such as temperature, humidity, and light; by aging; and by conservation treatments. 3 AGING OF PAPERThe primary concerns of research on the aging of paper involve determination of 1) the effects of natural aging on paper and 2) the parameter of accelerated aging that most nearly match the effect of natural aging on paper. 3.1 NATURAL AGINGNatural aging may cause paper to become discolored and cellulose to become more crystalline and stiff, as has been shown for accelerated aging (fig. 5). Crystallinity can be measured by x-ray diffraction, acid hydrolysis, or moisture content, although the percent crystalloids depends on the method used and on interference of additives (Daniels 1986). Koura and Krause (1987) concluded that, based on changes in mechanical properties, treatments using ammonia could reverse the degree of crystallinity in aged papers. Discoloration of a paper sheet may be caused by the formation of chromophores upon aging as a result of exposure to, among other things, light (Lee et al. n.d.) and volatile gases (Daniels 1988). Daniels has hypothesized that oxidation can be induced in paper by hydrogen peroxides from oxidation of lignin, leading to autoxidation of paper (Daniels 1988). He has described how hydrogen peroxide can be recorded by the Russell effect, which produces an image on sensitive photographic film. More recent studies have focused on the effects of pollution on aging of papers, particularly deacidified papers, which appear to have a greater capacity to absorb sulfur dioxide and nitrogen dioxide (G.C.I. Newsletter 1990).
3.2 ACCELERATED AGINGAccelerated aging, undertaken at elevated temperatures and humidities, may initiate reactions resulting from thermal degradation. These reactions could, depending on conditions and length of exposure, produce products different from those of natural aging (Feller 1987). Erhardt has used gas chromatography mass spectrometry to analyze extracts of papers aged at different temperatures and relative humidities. He found that humid oven aging of Whatman #1 filter paper (at 90�C and 100% RH) hydrolyzed cellulose and xylan, producing mostly glucose and xylose, also found in naturally aged papers. Dry oven aging at 90�C and above produced very little glucose. Aging at 150�C produced a different set of products (Erhardt et al. 1987; Erhardt 1988). Other researchers have focused on the effects of cycling aging conditions, finding that cycling relative humidity increases the degradation of single sheets of paper (Shahani et al. 1989). However, the effects of cycling appear to be less pronounced for books, the format of which may slow down changes. On the other hand, simulated book materials tend to age faster in comparable accelerated aging conditions than do single sheets, perhaps owing to the trapped concentration of degraded materials forming what amounts to an adverse microclimate. As noted by Erhardt (1988), there are many standards for accelerated aging applied to paper. For instance, TAPPI test methods lists three different sets of conditions ranging from 90–105�C and 0–100% RH depending on the type of papers and property to be measured, while ISO requires 80�C and 65% RH. In general, a wide variety of aging conditions continue to be used in current research, including the studies described in the following section. 4 CONSERVATION TREATMENTSThe investigations reported in this section address changes to paper properties caused by various treatments. Conservators are cautioned to remember that the demands of good research limit the scope of experimental design, which cannot possibly be expected to incorporate all conditions of actual conservation treatment practice. The practicing conservator should recognize that reported data pertain to specific experimental conditions. However, the data may be correlated to actual practice by extrapolation based on an understanding of the interaction of paper properties. For instance, one might hypothesize that a bleach found to damage cellulose in a cotton paper may also damage cellulose in a paper made of more reactive ground wood pulp. By the same token, changes to properties caused by one solvent application technique may not occur with an alternate application technique. The conservator must also recognize that the interpretation of research data requires great care. Treatments cause changes to more than one property. Some property changes may be perceived as beneficial while others may be considered detrimental, and indeed, these initial assessments may be reversed over time as the object ages. Each treatment of an object must be considered individually to decide whether the net benefits outweigh the disadvantages, depending on the needs and character of that particular object. For example, washing might remove discoloration, but this benefit must be balanced against a loss or change in sizing. Sizing agents such as gelatin impart strength to paper (Barrett 1989). A loss of sizing might reduce breaking length by decreasing tensile strength. At the same time, a loss of sizing might improve fold endurance by decreasing stiffness. The changes in properties may or may not be relevant, depending on subsequent conservation treatment (i.e., resizing) or the needs of the object. For instance, an improvement in fold endurance might be more beneficial for paper used in a book than in flat artwork. Furthermore, experimental conditions must be carefully considered when comparing data from different projects. Minor variations, for instance in techniques of sample preparation involving wetting or drying, may inadvertantly affect properties of gloss or tensile strength. Modifications in degree of solvent saturation and direction of application, or in drying time, pressure, and technique, must be taken into account. All of these variables are factors that a conservator must consider when formulating treatment options, particularly regarding the most intrusive conservation treatments of all: those involving washing or deacidification, bleaching, solvents, enzymes, and sizing. 4.1 WASHINGWashing in paper conservation refers to overall treatment of paper with aqueous solutions, usually containing additives (such as wetting agents, solvents, or alkaline compounds) that serve to swell paper fibers and dislodge or dissolve impurities in order to aid in cleaning, neutralize acidity, or deposit an alkaline reserve to guard against future acid exposure (Daniels and Shashoua 1991). Early research found that washing with deionized or distilled water could cause a drop in pH and fold endurance, since such highly purified water can dissolve the stabilizing magnesium and calcium components of paper (Tang and Jones 1979; Nelson et al. 1982). On the other hand, alkalinization, either with low concentrations used for cleaning or higher concentrations used to deacidify paper, may cause color change, especially in papers containing lignin, the unstable impurity in ground or mechanical wood pulp (Burgess et al. 1990). In order to differentiat!#e papers that may be improved with selected washing techniques from those that may not, Burgess has analyzed naturally aged papers that were washed in deionized water and in water containing low (20 ppm) and high (200 ppm) concentrations of magnesium compounds, before and after humid thermal accelerated aging. Washing with deionized water did remove some beneficial calcium and magnesium salts. However, since washing also removed acid impurities, sample papers with high initial acidity that were washed actually maintained after aging a degree of polymerization greater than the unwashed untreated controls, which underwent more acid hydrolysis during aging. On the other hand, the degree of polymerization for papers with low initial acidity did indeed drop, relative to the controls, after washing with deionized water followed by accelerated aging. In addition, although some alkaline-treated, lignin-containing papers maintained greater mechanical strength with zero-span tensile measurements, an increase in concentration of some alkaline magnesium components produced an increase in yellowing of the lignin-containing papers. As a result, the authors concluded that, pending further studies on lignin-containing papers, institutions should “consider reducing the quantity of ground wood papers which are being treated by aqueous deacidification,” shifting priorities to materials with little or no lignin (Burgess et al. 1990). Increase in yellowing may also occur in ligneous ground or mechanical wood pulp treated by solvent immersion (Lienardy and van Damme 1990). Ligneous paper washed with 30% isopropanol in tap water registered a drop in reflectance as compared to the control (fig. 6a). For all wood pulp paper samples, immersion for 5 minutes in alcohol (followed by washing in water for 30 minutes), caused an increase in fold endurance but a reduction in breaking length (fig. 6b and fig. 6c). Breaking length is the calculated upper limit of length of a uniform paper strip that would support its own weight if it were suspended at one end. It is a convenient measure for comparing the tensile breaking strength of papers of varying grammages (Caulfield and Gunderson 1988). A similar effect of alcohol was found in an earlier study, in which immersion of a mixed cottonwood pulp, alum rosin sized paper in 9:1 ethanol:deionized water appeared to cause a slight reduction in elongation to break (fig. 7a and fig. 7b) as compared to samples immersed in water alone (van der Reyden et al. 1988). (Reprinted with permission from from van der Reyden et al. 1988)
Reductions in breaking length, or in elongation to break, could result from a breakdown of sizing or the disruption of hydrogen bonding in paper immersed in organic solvent, which may in effect dehydrate or debond the paper. On the other hand, immersion in water swells fibers and releases the strain built into paper during the original drying of the sheet and subsequent aging. Aging stiffens paper as the random array of cellulose chains in the flexible amorphous regions of fibrils become more ordered and crystalline, held rigid by hydrogen bonds between the cellulose chains (fig. 2). Washing introduces water molecules that sever these bonds in the amorphous regions (fig. 3). Water can act as a plasticizer, swelling fibers and allowing fibrillar rearrangements that build in creep and encourage rebonding at less stressed sites upon drying. This phenomenon may account in part for the increase in stretch or elongation to break after washing (unsized) paper in alkaline solutions (fig. 4). Stretch is regarded by some in the paper industry as “one of the most important criteria for satisfactory behavior of paper in use” (Higgins and de Yong 1962). Furthermore, “a stress-strain curve in which the initial slope (Young's modulus) is high, where there is little curvature, and where the value for elongation to break is low, indicates a brittle material. A tough material is one that exhibits more viscoelastic behavior, a pronounced curvature, and a high elongation to brea” (Caulfield and Gunderson 1988). 4.2 BLEACHINGBleaching, potentially damaging to paper and fibers, is nonetheless occasionally used by some paper conservators when washing fails to remove stains. Many paper conservators have become particularly interested in aqueous light bleaching, which consists of bleaching paper immersed in an alkaline solution by exposing it to light (Keyes 1980). Feller investigated the role of pH in aqueous light bleaching and found little difference in bleaching efficiency among solutions with pH 7–10, either before or after aging, on thermally discolored filter paper (fig. 8a) with little lignin or hemicellulose content (Lee et al. n.d.). He also found insignificant photochemical damage as manifested by color reversion or change in degree of polymerization after accelerated thermal aging of aqueously light bleached filter paper samples (fig. 8b). This parallels findings regarding mechanical properties of an earlier study (van der Reyden et al. 1988), which has been expanded to include accelerated aging (Schaeffer et al. n.d.). The tensile strength of cotton-fibered Whatman #1 paper aqueously light-bleached with UV filtration in an Atlas Weather-Ometer is statistically identical to all washed controls, with an increase in elongation remaining greater after aging than the aged untreated control (fig. 9a, fig. 9b). On the other hand, a naturally aged artists' paper with similar fiber composition,. but heavily sized with alum and gelatin underwent a reduction in stress to failure or load to break for samples immersed in a solution of calcium hydroxide (pH 9.5) in the Weather-Ometer (fig. 9c, fig. 9d). This situation is probably the result of a breakdown in sizing, especially when the paper is subjected to thermal degradation during aqueous light bleaching in the (warm) Weather-Ometer. The reduction in strain, or elongation to break, before aging might be attributable to thermal or photooxidation of the paper or sizing. However, after aging the strain measurements of both types of immersed samples are statistically identical to each other. Interestingly, the aqueously light-bleached samples underwent the least color reversion of all samples, particularly in yellowing, as measured by CieL∗a∗b∗ (fig. 9e). Lienardy and van Damme (1990) reported similar results for samples aqueously bleached by unfiltered sun and artificial light, although the samples underwent a drop in fold endurance, which may indicate ultraviolet degradation and/or a breakdown or alteration in the sizing of their naturally aged test papers. Naturally aged test papers pose several problems with respect to good research design since such papers vary considerably in fiber, size, additives, and degradation products even within a single sheet. Such papers may also have low degrees of polymerization and tensile strength, which make changes difficult to measure. Consequently, naturally aged papers should be tested along with controls of similar fiber composition, which can be thermally pre-aged, and may provide the outer parameters for treatment evaluation. If relatively strong papers (such as the filter and artists' papers made by Whatman) are damaged by a treatment, naturally aged papers of lesser quality may be expected to be damaged as well. The effects of aqueous light bleaching have been compared to those of chemical bleaches (van der Reyden et al. 1988; Lienardy and van Damme 1989; Hofmann et al. 1990; Flieder et al. 1991). Findings before and after aging indicate that aqueous light bleaching, with UV filtration, causes no significant damage to the mechanical strength of a preaged cotton paper as compared to chemical bleaches (fig. 10) when all bleaching conditions are followed by extensive washing (Hofmann et al. 1992). Regarding chemical bleaches in general, Burgess (1990a) has shown that although both hydrogen peroxide and sodium borohydride produce excellent color stability with respect to reversion, increasing the pH of stabilized hydrogen peroxide to pH10 caused a decrease in the degree of polymerization by 21% in a 19th-century rag paper. However, sodium borohydride can actually stabilize damaged cellulose, reducing the color-producing double bonds of two types of carbonyls (aldehydes and ketones) back to original colorless alcohol species (Burgess 1988).
4.3 SOLVENTSUse of solvents can often preclude the need to bleach, by removing stains and adhesive residues, but paper conservators remain concerned about the effects of solvents on paper (MacKay and Smith n.d.). One problem, for instance, is that local treatments can sometimes produce tidelines which are only visible in ultraviolet light, or after aging of the treated object. In addition to the publications mentioned in the washing section, recent studies have focused on particularly solvent sensitive papers such as modern chemical watermarked papers (Watkins 1990), historic transp35arent paper (Flieder et al. 1988; Richardin et al. 1990), and coated papers (Baker et al. 1989). Such papers are affected by both choice of solvent agents and choice of application technique. Recent work has shown that choice of solvent diluent (such as ethanol, acetone, or toluene), affects properties of selected polymers (Hansen et al. 1991). A study comparing solvents and application techniques for stain removal (using immersion, suction table, and poultice) has documented alteration of surface fin46ish of paper, including break-up of impregnants, leading to a loss of translucency in tracing papers (fig. 11). The effects of solvents on coated papers parallel in some ways findings by paintings conservators on the reaction of pigment films to solvent swabbing and immersion (Hedley et al. 1990). For example, application of aqueous poultices to some coated papers may produce what appear to be tideline stains but are actually cracks (fig. 12). Attempts to reduce stains on coated papers with solvents could actually increase cracking (van der Reyden n.d.). Clearly, there is a need to continue to characterize specialty papers using Fourier transform infrared spectroscopy, gas chromatography/mass spectrography, and scanning electron microscopy/energy dispersive spectrometry in order to track effects of alternative solvent application techniques with SEM dot mapping and UV reflectance/transmission microscopy.
4.4 ENZYMESLike many other conservators, paper conservators employ enzymes to break down adhesives among other things. Burgess (1990b) has reported on criteria for enzyme selection and on the discrepancies between the working properties and information found in specifications on manufacturers' labels. Labels may record enzyme activity measured on substrates such as casein (which is different from adhesives generally encountered by paper conservators), as well as the pH at which the enzyme was assayed, rather than the pH for optimum activity (Grattan et al. 1987). In fact, some enzymes are biphasic, having high activity levels in both acid and basic pH ranges (fig. 13). Although enzyme activity approximately doubles in rate for each 10�C rise from 20–60�C, activity can be increased at lower temperatures by increasing the concentration of enzyme. However, another study, in which amylase enzymes were tagged with radioactive iodine isotopes, indicated that even though most of the enzymes were removed by rinsing the test papers twice with water, the amount of enzyme residue remaining in the paper after rinsing, although quite small, increased with an increase in concentration and purity (Andrews et al. 1990). The authors therefore recommended that conservators use the minimum amount of enzyme needed to do the job.
4.5 SIZINGPaper conservators are concerned about whether any of the solutions discussed above alter or remove the sizing in paper, whether papers should be resized, and if so, with what (Barrett 1989). Barger has examined papers, from 17 disbound books, which after washing and deacidification were resized with gelatin, parchment, wheat starch, methylcellulose, and Klucel-G (Carapella et al. 1990). SEM examination revealed that the surface relief of the papers selected for treatment played a large role in the conformation and adhesion of sizing. For instance, gelatin adhered more poorly to high-surface-relief rag papers than to more planar wood pulp paper. The sizes tested did not appear to undergo significant morphological change after accelerated aging, aside from some curling and cracking of gelatin size. However, Feller's study of cellulose ethers found poor color stability, after accelerated aging, for hydroxypropyl cellulose powders like Klucel-G, with a 28% decrease in reflectance (Feller and Wilt 1990). Hydroxypropyl cellulose powders appear to be more susceptible to thermal degradation than other cellulose ethers because of a 60% degree of substitution of hydroxyls by etherifying reagents. Feller's study also found that in general, cellulose ethers with lower molecular weights tend to discolor more than those with higher molecular weights. 5 CONCLUSIONEvaluating conservation research is especially challenging because of the diversity in research design, test conditions and procedures, and the interpretation of results and recommendations. The importance of good research design—requiring a limited area of investigation, reduction of variables, thorough characterization of samples, and familiarization with the chemistry of materials in order to understand the advantages and disadvantages of selected test procedures—cannot be overemphasized (Burgess and Binnie 1990). On the other hand, procedures are continually being developed and refined to improve instrument sensitivity and the quality of information, and this may account for apparent inconsistencies in research. For instance, there is a need to codify accelerated aging conditions such as temperature, relative humidity, time, sample mounting techniques, and air-to-sample volume ratios, to determine the best modes for color measurements (e.g., the use of whiteness, brightness, reflectance, or CieL∗a∗b∗ or Yxy modes, relative to standard or target values, and recorded as difference or absolute measurement); and to further improve and adapt chemical and mechanical tests. Consequently, because of the constant evolution of procedures, it is imperative that all data be recorded and available for reinterpretation. Conservators must evaluate conclusions and recommendations in the context of the specifics of a particular research project, and conservators alone bear the responsibility for determining how findings apply to their own special cases. Researchers can supply only increments of information, which often generate more questions, and research findings can only aid, not replace, a conservator's good judgment. ACKNOWLEDGEMENTSI would like to thank the authors and speakers who allowed their work to be summarized in this article and the AIC Conservation Science Task Force, whose survey of research areas of interest to paper conservators reflected many of the topics covered. I would also like to acknowledge the numerous other conservators, interns, and conservation scientists who shared the following information about ongoing projects at their institutions. The research team headed by Chandru Shahani at the Library of Congress continues with projects on mass deacidification with diethyl zinc, the optimum alkaline reserve for paper, and the amount deposited by aqueous versus nonaqueous deacidification methods as well as on the diffusion of gases and acids in polyester film encapsulation and the effect of aging specimens in polyproylene enclosures as compared to humid aging. Susan Lee Bechtold reports that the National Archives is researching stable plastic enclosures for shrink wrapping damaged text blocks as well as the aging characteristics of adhesives, paper, film, tapes, and disks. At the Conservation Analytical Laboratory, Smithsonian Institution, Timothy Vitale is investigating the effects of drying on paper, having organized a seminar including work by Jon Arney, who is developing a system to measure changes in surface texture of paper. At the National Gallery of Art, Shelley Fletcher and Elmer Eusman are expanding on Pia De Santis's exploration of solvent residues and brown-line effects in naturally aged papers. The effect of aqueous light bleaching on sized rag paper is being continued by Terry Schaeffer as a collaborative effort with the Los Angeles County Museum of Art and the Getty Conservation Institute. At the Art Conservation Program, State University College at Buffalo, Irene Br�ckle is investigating the effects after aging of increasing concentrations of aluminum sulfate in gelatin sized papers. Finally, I would like to thank the many interns and colleagues who participated in projects mentioned above or reviewed and critiqued the manuscript, including Konstanze Bachman, Katherine Eirk, Eleanor McMillan, Nancy McRaney, Alan Postlethwaite, Olga Souza, Chris Tahk, Charles Tumosa, and Lambertus van Zelst. REFERENCESAndrews, T. M., W. W.Andrews, and C., Baker. 1990. An investigation into the removal of enzymes from paper following conservation treatment. American Institute for Conservation Book DFand Paper Group Annual9:1–11. Baker, M., D.van der Reyden, and N.Ravenel. 1989. FTIR analysis of coated papers. American Institute for Conservation Book and Paper Group Annual8:1–12. Barrett, T.1989. Early European papers/contemporary conservation papers: A report on research undertaken from fall 1984 through fall 1987. Paper Conservator13:1–108. Berndt, H.1989. A reexamination of paper yellowing and the Kubelka-Munk theory. In HistorEGic Textile and Paper Materials II: Conservation and Characterization, ed.S. H.Zeronian and H. L.Needles. American Chemical Society Symposium Series no. 410. Washington, D.C.:ACS.81–93. Bolam, F., ed.1962. The formation and structure of paper: Transactions of the symposium held at Oxford September 1961, vols. 1 and 2. London: Technical Section, British Paper and Board Makers' Association. Bolam, F., ed.1976. The fundamental properties of paper related to its usFHes: Transactions of the symposium held at Cambridge, September 1973, vols. 1 and 2. London: Technical Section, British Paper and Board Makers' Association. Burgess, H.1983. Chemistry of cellulose degradation. Lecture presented at AIC Book and Paper Specialty Group Annual Meeting. Burgess, H.1988. Practical considerations for conservation bleaching. Journal of the International Institute for Conservation–Canadian Group13:11–26. Burgess, H.1990a. The stabilization of cellulosic fibres by borohydride derivatives. ICOM Committee for Conservation preprints: 9th Triennial Meeting, Dresden. 447–52. BurgessH.1990b. An overview of enzymes: The chemistry of proteins and enzymes, working properties and how to select the appropriate enzymes for specific conservation treatments. Paper distributed at conference on Enzyme Treatments: The Science and the Applications in Conserving Artistic/Historic Works, Cambridge, Mass. Burgess, H. D., and N. E.Binnie. 1990. The development of a research approach to the scientific study of cellulosic and ligneous materials. Journal of the American Institute for Conservation29:132–52. Burgess, H., S.Duffy, and S.Tse. 1990. Investigation of the effect of alkali on paper. American Institute for Conservation Book and Paper Group Annual9:29–36. BurgessH. D., and N. E.Binnie. 1991. The effect of Vikane™ on the stability of cellulosic and ligneous materials: Measurement of deterioration by chemical and physical methods. In Materials Issues in Art and Archaeology II, Materials Research Society Proceedings no. 185. ed.P.Vandiver. et al 791–98. Carapella, C., E.Powell, E.Rutiser, and S.Barger. 1990. Changes in paper strength morphology caused by paper resizing treatment. Restaurator11:219–35. Casey, J., ed.1980. Pulp and paper: Chemistry and chemical technology. 3d ed.4 vols. New York: Wiley-Interscience. Caulfield, D. F., and D. E.Gunderson. 1988. Paper testing and strength characteristics. In TAPPI Proceedings: 1988 Paper Preservation Symposium 19–21 October, ed.R.Smith and T.Norris. Washington, D.C.: Technical Association of Pulp and Paper Industry. 31–40. Daniels, V.1986. Study of crystallinity of paper before and after paper conservation. Paper Conservator10:70–72. Daniels, V.1988. The discoloration of paper on aging. Paper Conservator12:93–101. Daniels, V., and Y.Shashoua. 1991. Wet cleaning of paper and textiles: Similarities and differences. In Paper and Textiles: The Common Ground, Preprints of the conference held at the Burrell Collection, Glasgow, 19–20 September 1991. The Scottish Society for Conservation and Restoration. 19–27. Dwan, A.1987. Paper complexity and the interpretation of conservation research. Journal of the American Institute for Conservation26:1–17. Erhardt, D.1988. Paper degradation: A comparison of industrial and archival concerns. In TAPPI Proceedings: 1988 Paper Preservation Symposium 19–21 October, ed.R.Smith and T.Norris. Washington, D.C.: Technical Association of Pulp and Paper Industry. 83–90. Erhardt, D., D.von Endt, and W.Hopwood. 1987. The comparison of accelerated aging conditions through the analysis of extracts of artificially aged papers. AIC preprints, 15th Annual Meeting, American Institute for Conservation, Washington, D.C.43–55. Feller, R.1987. Some factors to be considered in accelerated aging tests. AIC preprints, 15th Annual Meeting, American Institute for Conservation, Washington, D.C.56–67. Feller, R., and M.Wilt. 1990. Evaluation of cellulose ethers for conservation. Malibu, Calif.: Getty Conservation Institute. Flieder, F., F.Leclerc, C.Laroque, P.Richardin, and B.Guineau. 1988. Analyse et restauration des papiers transparents anciens. In Les documents graphiques et photographiques, analyses et conservation 1986–1987. Paris: Archives Nationales. 93–139. Flieder, F., M.Leroy, J. C.Andreoli, and F.Leclerc. 1991. Comparative study of four paper bleaching methods. In Proceedings for the International Federation of Library Associations–International Preservation and Conservation Research Seminar, ed.D.Clements. New York: IFLA. G.C.I. Newsletter. 1990. Exposure of deacidified paper to sulfur dioxide and nitrogen dioxide. Getty Conservation Institute Newsletter5(2):6. Grattan, D., J.St. Hilaire, H.Burgess, and J. C.McCawley. 1987. The use of enzymes in partially non-aqueous media. In Conservation of Library and Archives Materials and the Graphic Arts, ed.G.Petherbridge. London: Butterworths. 15–24. Hansen, E., M.Derrick, M.Schilling, and R.Garcia. 1991. The effects of solution application on some mechanical and physical properties of thermoplastic amorphous polymers used in conservation: Poly(vinyl acetate)s. Journal of the American Institute for Conservation30:203–14. Hedley, G., M.Odlyha, A.Burnstock, J.Tillinghast, and C.Husband. 1990. A study of the mechanical and surface properties of oil paint films treated with organic solvents and water. In CleQSaning, Retouching and Coatings, ed.J.S.Mills and P.Smith. London: IIC.98–105. Higgins, H. G., and J.de Yong. 1962. The beating process: Primary effects and their influence on pulp and paper properties. In The formation and structure of paper: Transactions of the symposium held at Oxford, September 1961, ed.F.Bolam. London: Technical Section, British Paper and Board Makers' Association. 2:651–97. Hofmann, C., D.van der Reyden, and M.Baker. 1992. Comparison RTand evaluation of bleaching procedures: The effect of five bleaching methods on the optical and mechanical properties of new and aged cotton linter paper before and after accelerated aging. American Institute for Conservation Book and Paper Group Annual. 10:109–27. Hofmann, C., V.Flamm, G.Banik, and K.Messner. 1990. Bleaching of foxing stains in Art Nouveau. ICOM Committee for Conservation preprints, 9th Triennial Meeting, Dresden. 472–78. Hollinger, W.1988. Conservation Resources catalogue. Springfield, Va.: Conservation Resources International. Inaba, M., and R.Sugisita. 1991. Permanence of washi (Japanese paper). Part 2. In Materials Issues in Art and Archaeology II, Materials Research Society Proceedings no. 185, ed.P.Vandiver. et al 799–804. Keyes, K.1980. Alternatives to conventional methods of reducing discoloration in works of art on paper. In Preprints from the Cambridge 1980 International Conference on the Conservation of Library and Archive Materials and the Graphic Arts. Cambridge, Eng: IPC. 166–70. Koura, A., and T.Krause. 1987. Increase in paper permanence by treatment with liquid ammonia or ammonia solutions. Part 1, Fundamental basis and influence on fiber and paper structure and properties. In Conservation of Library and Archives Materials and the Graphic Arts, ed.G.Petherbridge. London: Butterworths. 31–36. Kremer, R., and D.Tabb. 1990. Paper: The beneficially interactive support medium for diagnostic test development. American Laboratory22(3):136–43. Kruth, L.1988. A survey of recent scientific research which has caused a re-evaluation of commonly used practises in book and paper conservation. American Institute for Conservation Book and Paper Group Annual7:30–39. Lee, S. B., J.Bogaard, and R. L.Feller. n.d. Bleaching by liVXght I: Effect of pH on the bleaching or darkening of papers in the dry and in the immersed condition under visible and near-ultraviolet radiation. Bleaching by light II: Studies of the bleaching of thermally-discolored sugars and other “model” compounds. In Symposium 88: The conservation of historic and artistic works on paper. Ottawa: Canadian Conservation Institute. Forthcoming. Lienardy, A., and P.van Damme. 1989. Resultants de recherches experimentales sur le blanchiment du papier. Studies in Conservation34:123–36. Lienardy, A., and P.van Damme. 1990. Paper washing. Paper Conservator14:23–29. Luner, P.1988. Evaluation of paper permanence. Wood Science and Technology22:81–97. MacKay, C., and A.Smith. n.d. The effect of wetting agents on the tensile strength of paper. In Symposium 88: The conservation of historic and artistic works on paper. Ottawa: Canadian Conservation Institute. Forthcoming. Nelson, J., A.King, N.Indictor, and D.Cabelli. 1982. Effects of wash water quality on the physical properties of three papers. Journal of the American Institute for Conservation18:59–76. Priest, D. J.1989a. Modern paper. In Modern art: The restoration and techniques of modern paper and paints, ed.S.Fairbrass and J.Hermans. London: United Kingdom Institute of Conservation. 5–7. Richardin, P., F.Flieder, S.Bonnaissies, and C.Pepe. 1990. Analyse par CY[G/SM des produits d'impregnation de papier calques anciens. ICOM Committee for Conservation preprints, 9th Triennial Meeting, Dresden. 482–88. Robb, A.1991. Selected bibliography on research in paper conservation since 1988. Unpublished manuscript. Washington, D.C.: Conservation Analytical Laboratory, Smithsonian Institution. Schaeffer, T. M.Baker, V.Blyth-Hill, and D.van der Reyden. n.d. Effect of aqueous light bleaching on the subsequent aging of paper. Journal of the American Institute for Conservation. Forthcoming. Shahani, C., F.Hengemihle, and N.Weberg. 1989. The effect of variations in relative humidity on the accelerated aging of paper. In Historic textile and paper materials II: Conservation and characterization, ed.S. H.Zeronian and H. L.Needles. American Chemical Society Symposium Series no. 410. Washington, D.C.: ACS.63–80. Smith, A.1991. Cellulose: In paper and textiles. In Paper and Textiles the Common Ground. Preprints of the conference held at the Burrell Collection, Glasgow, 19–20 September 1991. The Scottish Society For Conservation and Restoration. 1–17. Tang, L.C., and N.M.M.Jones. 1979. The effects of wash water quality on the aging characteristics of paper. Journal of the American Institute for Conservation18:61–81. van der Reyden, D., M.Mecklenburg, M.Baker, and M.Hamill. 1988. Update on current research into aqueous light bleaching at the Conservation Analytical Laboratory. American Institute for Conservation Book and Paper Group Annual7:73–106. van der Reyden, D. n.d. Some effects of solvents on transparent papers. Postprints of the Institute for Paper Conservation Conference, April 1–4, 1992. Forthcomin. Watkins, S.1990. Chemical watermarking of paper. Journal of the American Institute for Conservation29:117–31. Waterhouse, J. F.1988. Monitoring the aging of paper. In TAPPI Proceedings: 1988 Paper Preservation Symposium. ed.R.Smith and T.Norris. Washington D.C.: Technical Association of Pulp and Paper Industry. 59–63. Wilson, W. K., and E. J.Parks. 1979. An analysis of the aging of paper: Possible reactions and their effects on measurable properties. Restaurator (3) 1–2:37–61. Wozniak, J.1985. The use of ultrasonic measurements to monitor the aging of paper. Unpublished manuscript. Atlanta: Institute of Paper Science and Technology. OTHER SOURCESAtalla, R. H.1981. The crystallinity of cellulosic fibers. Dependence on history and influence on properties. In Preservation of Paper and Textiles of Historic Value II. Washington, D.C., ACS. 169–76. Cooper, D., C.King, and J.Segal. 1987. The use of enzymes in partially non-aqueous media. In Conservation of Library and Archives Materials and the Graphic Arts, ed.G.Petherbridge. London: Butterworths. 25–30. Erhardt, D.1989. Relation of reaction rates to temperature. Abbey Newsletter13:38–39. Flamm, V., C.Hofmann, S.Dobruskin, and G.Banik. 1990. Conservation of tracing papers. ICOM Committee for Conservation preprints, 9th Triennial Meeting, Dresden. 463–67. Flieder, F. n.d. Analysis and restoration of old transparent papers. In Symposium 88: The conservation of historic and artistic works on paper. Ottawa: Canadian Conservation Institute. Forthcoming. Futernick, R. n.d. New methods, adapted tools and alternate treatments. Symposium 88: The conservation of historic and artistic works on paper. Ottawa: Canadian Conservation Instititue. Forthcoming. G.C.I. Newsletter. 1988. Cellulose ethers show markedly different aging properties. Getty Conservation Institute Newsletter3(1):7. Hansen, E., and C.Reedy. 1991. Replies to AIC Task Force on Conservation Science Questionnaire to the Book and Paper Group. Unpublished manuscript. Hon, D.N.S.1989. Critical evaluation of mass deacidification processes for book preservation. In Historic Textile and Paper Materials II: Conservation and Characterization, ed.S. H.Zeronian and H. L.Needles. American Chemical Society Symposium Series no. 410. Washington, D.C.: ACS. 13–34. Keyes, K.1978. The unique qualities of paper as an abdrtifact in conservation treatment. Paper Conservator3:3–8. Kimberly, A., and J.F.C.Hicks. 1931. Light sensitivity of rosin paper-sizing materials. Bureau of Standards Journal of Research6:819–27. Kouris, M., ed.1983. Pulp and paper manufacture: Coating, converting, and specialty processes. 3d ed. Vol. 8. Lee, S. B., J.Bogaard, and R. L.Feller. 1989a. Damaging effects of visible and near-ultraviolet radiation on paper. In Historic Textceile and Paper Materials II: Conservation and Characterization, ed.S. H.Zeronian and H. L.Needles. American Chemical Society Symposium Series no. 410. Washington, D.C.: ACS.54–62. Lee, S. B., J.Bogaard, and R. L.Feller. 1989b. Darkening of paper following exposure to visible and near-ultraviolet radiation. Journal of the American Institute for Conservation28:1–18. Levison, H. W., F.Sutil, and E. T.Vanderbrink. 1987. Lightfastness of pigmented handmade papers. Color Research and Application12:37–41. Lienardy, A., and P.van Damme. 1988. A bibliographical survey of the bleaching of paper. Restaurator9:178–98. Mark, R. E.1983. Handbook of physical and mechanical testing for paper and paperboard. New York and Basel: Marcel Dekker. Mills, J., and R.White. 1987. The organic chemistry of museum objects. London: Butterworths. Morse, E.1990. Selected Bibliography. Paper distributed at coegnference on Enzyme Treatments: The Science and the Applications in Conserving Artistic/Historic Works, Cambridge, Mass. Postlethwaite, A.1991. Achieving integrated pest eradication with heightened sensitivities to collections, safety, and human toxicity. In Proceedings for the International Federation of Library Association International Preservation and Conservation Research Seminar, New York. Priest, D. J.1989b. Permanence and alkaline-neutral papermaking. In Historic textile and paper materials II: Conservation and characterization, ed.S. H.Zeronian and H. L.Needles. American Chemical Society Symposium Series no. 410. Washington, D.C.: ACS. 2–12. Rance, H. F.1982. Handbook of paper science. 2 vols. New York: Elsevier. Rapson, W. H., C. B.Anderson, and A.Magued. 1988. Natural aging of laboratory bleached pulps. In TAPPI Proceedings: 1988 Paper Preservation Symposium 19–21 October, ed.R.Smith and T.Norris. Washington, D.C.: Technical Association of Pulp and Paper Industry. 31–40. Ravines, P., N.Indictor, and D. M.Evetts. 1989. Methylcellulose as an impregnating agent for use in paper conservation. Restaurator10:32–46. Sebera, D.1990. Effects of strengthening and deacidification on paper permanence. Part 1: Some fundamental considerations. American Institute for Conservation Book and Paper Group Annuhjal9:65–117. Shahani, C. J., and W. K.Wilson. 1987. Preservation of libraries and archives. American Scientist75:240–51. Shelley, M.1989. Old master drawings: an approach to conservation. American Institute for Conservaiton Book and Paper Group Annual. 8:62–66. Shores, F.1987. A clean room for a small conservation laboratory. AIC preprints, 15th Annual Meeting, American Institute for Conservation, Washington, D.C.118–23. Siktone, J.1987. Treatment of 19th century tracing papers from the Frederick Law Olmsted Collection. Preprints, 8th Triennal Meeting, ICOM Committee for Conservation, Paris. 2:731–38. Tse, S., and H.Burgess. n.d. Effect of enzyme treatments on degradation of paper. In Symposium 88: The conservation of historic and artistic works on paper. Ottawa: Canadian Conservation Institute. Forthcoming. Wills, P., and N.Pickwood, eds.1985. Hyogu: The Japanese tradition jlin picture conservation. Paper Conservator9:1–63. FIGURE CREDITSTable 2 Reprinted with permission from Wilson, W. K., and E. J. Parks. 1979. An analysis of the aging of paper: Possible reactions and their effects on measurable properties. Restaurator (3) 1–2:37–61. Copyright 1979 by Munksgaard, 35 Norre Sogade, P.O. Box 2148, K-K 1016 Copenhagen K, Denmark. And with permission from Waterhouse, J. F. 1988. Monitoring the aging of paper. In TAPPI Prockmeedings: 1988 Paper Preservation Symposium. ed. R. Smith and T. Norris. Washington D.C.: Technical Association of Pulp and Paper Industry. 59–63. Figure 1a. Reprinted with permission from Dwan, A. 1987. Paper complexity and the interpretation of conservation research. Journal of the American Institute for Conservation 26:5. From Waterhouse, J. F. n.d. unpublished course notes. Figure 1b. Reprinted with permission from Hollinger, W. 1988. Conservation Resources catalogue. Springfield, Va.: Conservation Resources International. page 6. Figure 2. Reprinted with permission from Inaba, M., and R. Sugisita. 1991. Permanence of washi (Japanese paper). Part 2. In Materials Issues in Art and Archaeology II no. 185, ed. P. Vandiver et al. 799–804. Figure 3. Reprinted with permission from American Laboratory 22(3):142. Copyright 1990 by International Scientific Communications Inc. Figure 5. Reprinted with permission from Waterhouse, J. F. 1988. Monitoring the aging of paper. In TAPPI Proceedings: 1988 Paper Preservation Symposium. ed. R. Smith and T. Norris. Washington D.C.: Technical Association of Pulp and Paper Industry. 59–63. Figure 6. Figures x-y have been reproduced from Lienardy, A., and P. van Damme. 1990. Paper washing, The Paper Conservator 14:23–29 by permission of The Institute of Paper Conservation. Copies onpf this and other volumes are available from IPC, Leigh Lodge, Leigh, Worcestershire WR6 5LB, England, for �15 (U.S. $30) �10/$20 for IPC members. Figure 8. Reprinted with permission from Lee, S. B., J. Bogaard, and R. L. Feller. n.d. Bleaching by light I: Effect of pH on the bleaching or darkening of papers in the dry and in the immersed condition under visible and near-ultraviolet radiation. Bleaching by light II: Studies of the bleaching of thermally-discolored sugars and other “model” compooqunds. In Symposium 88: The conservation of historic and artistic works on paper. Ottawa: Canadian Conservation Institute. Forthcoming. Figure 13. Reprinted with permission from Grattan, D., J. St. Hilaire, H. Burgess, and J. C. McCawley. 1987. The use of enzymes in partially non-aqueous media. In Conservation of Library and Archives Materials and the Graphic Arts, ed. G. Petherbridge. London: Butterworths. 15–24. AUTHOR INFORMATIONDIANNE VAN DER REYDEN received an M.A. in arprt history (1979) and a diploma in conservation from the Conservation Center, Institute of Fine Arts, New York University (1980) and a certificate in paper conservation from the Harvard University Art Museums (1981), following internships at the Fogg Art Museum, the Library of Congress, and the Museum of Modern Art. She is a senior paper conservator and co-head of the Paper Conservation Laboratory at the Conservation Analytical Laboratory, Smithsonian Institution, engaged in training of interns and professionals and in research aimed at development and evaluation of paper conservation treatments. Address: Conservation Analytical Laboratory, Smithsonian Institution, Washington, D.C. 20560.
 Section Index Section Index |